Prescription 2017: Ashwagandha
February 21, 2017 Written by JP [Font too small?]The union of science and traditional medicine is a tricky business. The scientific method of identifying and objectively testing elements in food, medicinal plants and the environment is relatively new to the human experience. In generations past, observation and trial-and-error sufficed, and it still does for some populations and the majority of animals. Experiences are handed down from elders to the young and from non-scholastic healers to their communities. In some cases, advocates choose one of these philosophies over the other. But, increasingly, a middle ground is something both camps acknowledge may be the best of both worlds.
Ashwagandha (Withania somnifera Dunal) is one of the most revered remedies in the Indian healing tradition known as Ayurveda. The root of ashwagandha aka Indian ginseng or winter cherry has been used for thousands of years for a wide range of symptoms from fatigue to memory decline. It falls into a category of plant medicines that is referred to as adaptogens. As the name implies, adaptogens assist the body in adapting to various insults and/or stressors, thereby normalizing health and physiological function.
These days, you’re likely to find ashwagandha dispensed in capsules, liquid extracts or tablets. Use of the ground root or decoctions made from it is far less common. Admittedly, the modern forms are easier to take. However, changing the method of administration can sometimes affect the therapeutic potential. For this reason, a few companies are testing to verify that their modern versions of ashwagandha are as effective, if not more so, than the traditional forms. Two of the best example I know are described below.
KSM-66 sounds decidedly modern, even futuristic. In reality, the makers of this patented extract have succeeded in staying strikingly true to the source material. KSM-66 is a broad spectrum extract from organic roots of ashwagandha. The essence of the root is extracted without the assistance of alcohol or synthetic solvents. It differs from traditional use because it’s concentrated and standardized for a minimum of 5% withanolides – one of several “active” ingredients scientifically identified in ashwagandha. To date, there have been several peer-reviewed studies conducted on this product. The results indicate that KSM-66:
- Assists with weight loss by reducing food cravings, psychological stress and improving “mental well being”.
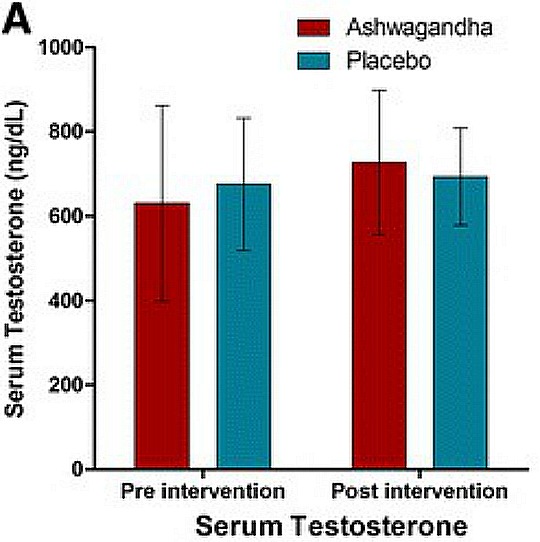
- Improves cardiorespiratory endurance and quality of life scores in healthy athletic adults. Other research has demonstrated greater muscle mass and strength gains when KSM-66 is combined with resistance training.
- Enhances sexual function and satisfaction in women and healthier hormone profiles and sperm quality in men.
- Lowers cortisol levels, a measure of stress, in adults living a high-pressure lifestyle.
General Dosage: The most common amount of KSM-66 used in the clinical research is 300 mg taken twice daily.
Sensoril, is another modern take on ashwagandha. It is a water-based extract that is derived from the leaves and roots of the plant. The use of the leaves is not typical. But, as it turns out, the leaves are a more abundant source of (beneficial) phytochemicals than the root. Extracting from both sources allows for this product to contain more than 10% Withanolide glycosides and 32% oligosaccharides. Several studies on this specific extract report that Sensoril:
- Reduces joint pain and disability without causing gastrointestinal disturbances.
- May improve cognitive and psychomotor performance in healthy adults. Other research supports the use of Sensoril in patients with bipolar-related cognitive dysfunction.
- Decreases the detrimental effects of acute and chronic mental stress on vascular function and related risk makers, such as blood pressure, C-reactive protein and elevated lipids.
- Improves endothelial function, while lowering inflammation and oxidative stress in type-2 diabetics.
General Dosage: The typical amount of Sensoril used in the studies is 250 mg twice daily.
In my opinion, both of these unique extracts stand out from the pack. Clearly, there are differences between the two in terms of exact composition and, even, price. KSM-66 tends to be less expensive, while Sensoril has a higher concentration of “actives”. What remains to be seen is how they’d perform if compared in a head-to-head trial. Is one more effective than the other? Which is safer? For instance, one study found that Sensoril can increase thyroxine (thyroid hormone) in some patients. This can be a positive thing in those who suffer from subclinical hypothyroidism and a negative “side effect” in those who don’t. In any event, some caution is warranted. Is the same true for those using KSM-66? I don’t have an answer to that. That’s why I always recommend informing your health care team whenever you decide to add a new supplement to your routine. That way, you will hopefully derive the benefits you seek while being certain that you’re not the recipient of unintended consequences.
Note: Please check out the “Comments & Updates” section of this blog – at the bottom of the page. You can find the latest research about this topic there!
To learn more about the studies referenced in today’s column, please click on the following links:
Study 1 – Effects of Ashwagandha (Roots of Withania Somnifera) on … (link)
Study 2 – Body Weight Management in Adults Under Chronic Stress Through … (link)
Study 3 – Efficacy of Ashwagandha (Withania Somnifera [L.] Dunal) in … (link)
Study 4 – Examining the Effect of Withania Somnifera Supplementation on … (link)
Study 5 – Efficacy and Safety of Ashwagandha (Withania Somnifera) Root … (link)
Study 6 – Clinical Evaluation of the Spermatogenic Activity of the Root Extract … (link)
Study 7 – A Prospective, Randomized Double-Blind, Placebo-Controlled Study … (link)
Study 8 – A Randomized, Double Blind Placebo Controlled Study of Efficacy … (link)
Study 9 – Evaluation of the Analgesic Activity of Standardized Aqueous Extract … (link)
Study 10 – Effect of Standardized Aqueous Extract of Withania Somnifera on Tests … (link)
Study 11 – Randomized Placebo-Controlled Adjunctive Study of an Extract of … (link)
Study 12 – Effect of Withania Somnifra Extract on Mental Stress Induced … (link)
Study 13 – A Comparative Study to Evaluate the Effect of Highly Standardized … (link)
Study 14 – Evaluation of a Highly Standardized Withania Somnifera Extract … (link)
Study 15 – A Standardized Withania Somnifera Extract Significantly Reduces … (link)
Study 16 – Subtle Changes in Thyroid Indices During a Placebo-Controlled Study … (link)
Ashwagandha + Resistance Trainig May Increase Testosterone in Men
Source: J Int Soc Sports Nutr. 2015 Nov 25;12:43. (link)
Tags: Ashwagandha, Cortisol, Stress
Posted in Diabetes, Diet and Weight Loss, Nutritional Supplements


February 21st, 2017 at 3:13 pm
Updated 02/21/17:
https://www.ncbi.nlm.nih.gov/pubmed/28207892
PLoS One. 2017 Feb 16;12(2):e0172508.
Triethylene glycol, an active component of Ashwagandha (Withania somnifera) leaves, is responsible for sleep induction.
Insomnia is the most common sleep complaint which occurs due to difficulty in falling asleep or maintaining it. Most of currently available drugs for insomnia develop dependency and/or adverse effects. Hence natural therapies could be an alternative choice of treatment for insomnia. The root or whole plant extract of Ashwagandha (Withania somnifera) has been used to induce sleep in Indian system of traditional home medicine, Ayurveda. However, its active somnogenic components remain unidentified. We investigated the effect of various components of Ashwagandha leaf on sleep regulation by oral administration in mice. We found that the alcoholic extract that contained high amount of active withanolides was ineffective to induce sleep in mice. However, the water extract which contain triethylene glycol as a major component induced significant amount of non-rapid eye movement sleep with slight change in rapid eye movement sleep. Commercially available triethylene glycol also increased non-rapid eye movement sleep in mice in a dose-dependent (10-30 mg/mouse) manner. These results clearly demonstrated that triethylene glycol is an active sleep-inducing component of Ashwagandha leaves and could potentially be useful for insomnia therapy.
Be well!
JP
February 21st, 2017 at 3:14 pm
Updated 02/21/17:
http://www.complementarytherapiesinmedicine.com/article/S0965-2299(16)30050-4/abstract
Complement Ther Med. 2016 Aug;27:25-9.
Evaluation of the efficacy of Withania somnifera (Ashwagandha) root extract in patients with obsessive-compulsive disorder: A randomized double-blind placebo-controlled trial.
BACKGROUND: Obsessive-compulsive disorder (OCD) is a chronic psychiatric disorder that is causally linked to dysregulation of the serotonergic system. The aim of this study is to investigate the efficacy of Withania somnifera (W. somnifera) root extract as an adjunct therapy to standard OCD treatment.
METHODS: Thirty patients with a confirmed diagnosis of OCD according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria participated in this randomized double-blind placebo-controlled trial and were randomly assigned to the treatment group (W. somnifera extract, 120mg/day; n=15) or the placebo group (n=15). All patients were under treatment with Selective Serotonin Re-uptake Inhibitors (SSRIs), and were instructed to take 4 capsules of the extract or placebo per day, preferably after meals, for a period of six weeks. The Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) was used in order to assess the severity of OCD symptoms at baseline and at the end of the trial. Statistical analyses were performed using SPSS software and Y-BOCS values were presented as median and range (Min-Max).
RESULTS: Comparison of the change in Y-BOCS score during the course of the trial revealed a significantly greater effect of W. somnifera (26 (14-40) [pre-treatment] versus 14 (4-40) [post-treatment]; change: -8 (-23 to 0)) versus placebo (18 (11-33) [pre-treatment] versus 16 (10-31) [post-treatment]; change: -2 (-4 to 0)) (P<0.001). The extract was safe and no adverse event was reported during the trial.
CONCLUSION: W. somnifera extract may be beneficial as a safe and effective adjunct to SSRIs in the treatment of OCD.
Be well!
JP
February 21st, 2017 at 3:18 pm
Updated 02/21/17:
http://link.springer.com/chapter/10.1007%2F978-3-319-41334-1_14
Adv Exp Med Biol. 2016;928:329-373.
Natural Withanolides in the Treatment of Chronic Diseases.
Withanolides, and in particular extracts from Withania somnifera, have been used for over 3,000 years in traditional Ayurvedic and Unani Indian medical systems as well as within several other Asian countries. Traditionally, the extracts were ascribed a wide range of pharmacologic properties with corresponding medical uses, including adaptogenic, diuretic, anti-inflammatory, sedative/anxiolytic, cytotoxic, antitussive, and immunomodulatory. Since the discovery of the archetype withaferin A in 1965, approximately 900 of these naturally occurring, polyoxygenated steroidal lactones with 28-carbon ergostane skeletons have been discovered across 24 diverse structural types. Subsequently, extensive pharmacologic research has identified multiple mechanisms of action across key inflammatory pathways. In this chapter we identify and describe the major withanolides with anti-inflammatory properties, illustrate their role within essential and supportive inflammatory pathways (including NF-κB, JAK/STAT, AP-1, PPARγ, Hsp90 Nrf2, and HIF-1), and then discuss the clinical application of these withanolides in inflammation-mediated chronic diseases (including arthritis, autoimmune, cancer, neurodegenerative, and neurobehavioral). These naturally derived compounds exhibit remarkable biologic activity across these complex disease processes, while showing minimal adverse effects. As novel compounds and analogs continue to be discovered, characterized, and clinically evaluated, the interest in withanolides as a novel therapeutic only continues to grow.
Be well!
JP
February 26th, 2017 at 6:12 pm
Updated 02/26/17:
http://www.tandfonline.com/doi/full/10.1080/19390211.2017.1284970
Journal of Dietary Supplements – Published Online 21 Feb 2017
Efficacy and Safety of Ashwagandha (Withania somnifera (L.) Dunal) Root Extract in Improving Memory and Cognitive Functions
Objectives: Cognitive decline is often associated with the aging process. Ashwagandha (Withania somnifera (L.) Dunal) has long been used in the traditional Ayurvedic system of medicine to enhance memory and improve cognition. Aim: This pilot study was designed to evaluate the efficacy and safety of ashwagandha (Withania somnifera (L.) Dunal) in improving memory and cognitive functioning in adults with mild cognitive impairment (MCI). Methods: A prospective, randomized, double-blind, placebo-controlled study was conducted in 50 adults. Subjects were treated with either ashwagandha-root extract (300 mg twice daily) or placebo for eight weeks. Results: After eight weeks of study, the ashwagandha treatment group demonstrated significant improvements compared with the placebo group in both immediate and general memory, as evidenced by Wechsler Memory Scale III subtest scores for logical memory I (p = 0.007), verbal paired associates I (p = 0.042), faces I (p = 0.020), family pictures I (p = 0.006), logical memory II (p = 0.006), verbal paired associates II (p = 0.031), faces II (p = 0.014), and family pictures II (p = 0.006). The treatment group also demonstrated significantly greater improvement in executive function, sustained attention, and information-processing speed as indicated by scores on the Eriksen Flanker task (p = 0.002), Wisconsin Card Sort test (p = 0.014), Trail-Making test part A (p = 0.006), and the Mackworth Clock test (p = 0.009). Conclusions: Ashwagandha may be effective in enhancing both immediate and general memory in people with MCI as well as improving executive function, attention, and information processing speed.
Be well!
JP